Zydus Cadila gets USFDA nod to market Potassium Chloride capsules
The company has more than 254 approvals and has so far filed over 350 abbreviated new drug applications (ANDAs) since it commenced filings in 2003-04.
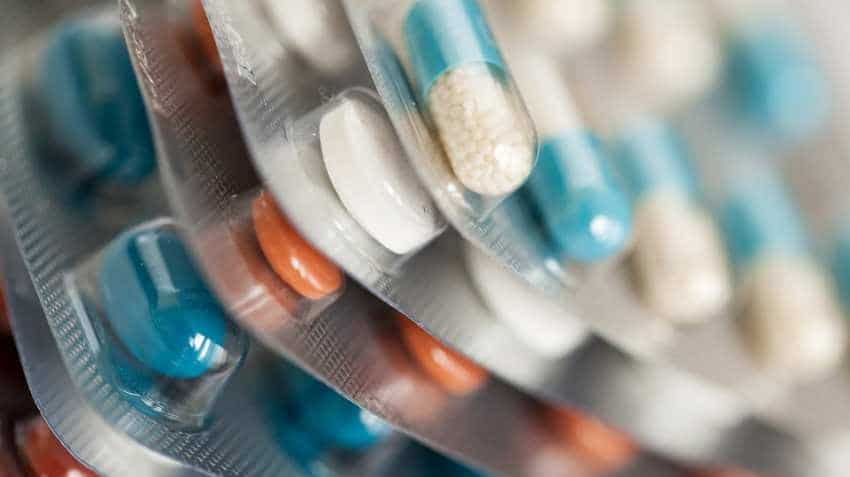
Drug firm Zydus Cadila Wednesday said it has received final approval from the US health regulator to market Potassium Chloride extended-release capsules, used to treat or prevent low amounts of potassium in the blood.
Zydus Cadila said the newly-approved product will be manufactured at the group's formulations manufacturing facility at SEZ, Ahmedabad.
The company has received approval from the US Food and Drug Administration (USFDA) to market the drug in the strengths of 8 mEq (600 mg) and 10 mEq (750 mg), Zydus Cadila said in a BSE filing.
This medication is a mineral supplement used to treat or prevent low amounts of potassium in the blood.
Potassium helps your cells, kidneys, heart, muscles and nerves work properly. Some conditions that can lower the body's potassium level include severe prolonged diarrhoea and vomiting, hormone problems such as hyperaldosteronism, or treatment with water pills/diuretics.
The company has more than 254 approvals and has so far filed over 350 abbreviated new drug applications (ANDAs) since it commenced filings in 2003-04.
Shares of the company's listed entity Cadila Healthcare were trading 1.07 per cent down at Rs 332.75 apiece on the BSE.
01:04 PM IST






 Zydus receives tentative approval from USFDA for Tofacitinib tablets
Zydus receives tentative approval from USFDA for Tofacitinib tablets Market Opening: Sensex rises 200 pts, Nifty by 60 pts on the last day of 2018
Market Opening: Sensex rises 200 pts, Nifty by 60 pts on the last day of 2018  Zydus Wellness rises over 4% as CCI clears deal with Heinz India
Zydus Wellness rises over 4% as CCI clears deal with Heinz India Zydus Cadila gets USFDA nod to market generic HIV treatment tablets
Zydus Cadila gets USFDA nod to market generic HIV treatment tablets